Down-flowing FCC reactor increases propylene, gasoline make
A 0.2-b/d fluid catalytic cracking unit (FCCU) using a downer reactor has demonstrated the ability of its high-severity process to enhance gasoline and light olefins production.
The process, jointly developed by the Petroleum Energy Center (PEC), Tokyo, and the King Fahd University of Petroleum and Minerals (KFUPM), Dhahran, boasts high catalyst-to-oil (C/O) ratios and a short contact time. Despite high operating temperatures, this configuration prevents back-mixing and suppresses thermal-cracking reactions, thereby maximizing gasoline and light olefins formation.
The performance of the downer-reactor system was studied in a modified Davison Circulating Riser (DCR; licensed by Grace Davison, Columbia, Md.) plant. In January and February 2000, the university conducted the test runs at three different C/O ratios; each run was operated for 6 hr after reaching steady state.
For comparative purposes, Petroleum Energy Center researchers at Nippon Mitsubishi Oil Corp., Tokyo, also operated a conventional riser-pilot plant at similar reactor conditions.
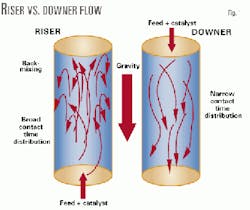
Fig. 1 illustrates the difference between downer and riser flows.
At 80 wt % conversion, the downer system had 22% more gasoline production than the riser system. Although the production of C2-C4 light olefins in the downer was high at 23 wt %, it was lower than light olefins production from the riser as a result of gasoline overcracking in the riser.
Yields of undesirable dry gas, coke, and 1,3 butadiene in the downer reactor were much lower. The results confirmed reduced thermal cracking and back-mixing in the downer-system configuration.
Based on these results, the two partners are designing a 30 b/d demonstration unit, to be built in 2002 near a Saudi refinery.
How to maximize yields
The increasing demand for light olefins, mainly propylene, for petrochemical feedstock1-4 has driven many FCCU operators to maximize their yields. The production of petrochemical feedstocks is economically attractive for refineries integrated with petrochemical plants.
Light olefins are also used to make reformulated-gasoline blending components such as methyl tertiary butyl ether (MTBE), tertiary amyl methyl ether (TAME), and alkylate.
About 14 million tonnes/year (or 30% of world supply) of propylene are a by-product of the FCCU. About 90% of this FCCU propylene goes to alkylate production.5
Generally, companies use three operating philosophies to maximize light olefins in conventional FCC units:5-7
- Increase riser-outlet temperature (increase severity). A conventional FCCU typically operates at low to moderate severity (510-530° C. riser outlet) with the flexibility to swing between maximum gasoline and maximum distillate modes.
Attempts to operate these units at higher severities to produce more light olefins will come at the expense of secondary cracking of gasoline, resulting in excessive dry gas (C2 and lighter) yields.
- Operate at short contact time. Short contact times reduce second-order polymerization reactions and allow less time for hydrogen-transfer reactions, thus enhancing olefins production. The use of this approach in conventional FCCU reactors (risers), however, will hinder the conversion of heavy oil fractions to light fractions.
- Use special catalysts or pentasil-type additives: Special FCC catalysts increase light olefins in conventional riser reactors. In general, these catalysts are prepared by increasing the ratio of matrix cracking over zeolite cracking and by using dealuminated zeolites (low-rare earth content).
ZSM-5 type additives are widely used to minimize hydrogen-transfer reactions and enhance gasoline octane and light olefins formation.
High-severity FCC
The objective of the new high-severity FCC (HS-FCC) process is to increase petrochemical feedstock production and final product yields.
The HS-FCC process uses a down-flow reactor to obtain a high reaction temperature (above 550° C.), short contact time, and a high C/O ratio. An efficient product separator suppresses side reactions (oligomerization and hydrogenation of light olefins) and coke formation accelerated by condensation.
- Down-flow reactor. The catalyst and gas flow downward with gravity to suppress back-mixing in the reactor and obtain a narrower distribution of residence time. The downward flow permits a higher C/O ratio, because the lifting of catalyst by vaporized feed is not required. The selection of an optimum residence time maximizes intermediate products such as gasoline and light olefins.
- Short contact time. Short contact times (less than 1 sec) minimize the effects of secondary reactions, reducing overcracking. They also maximize gasoline and light olefins yields.
- C/O ratio. A high C/O ratio maintains heat balance and helps restrain thermal cracking, overcracking, and hydrogen transfer.
Increasing the C/O ratio minimizes the effects of operating at high reaction temperature (thermal cracking). It increases conversion and decreases temperature drop during the endothermic cracking reaction.
Experiment
Fig. 2 shows a schematic of the downer-reactor plant. The pilot plant includes a catalyst hopper, feed section, downer-type reactor, catalyst stripper, spent-catalyst regenerator, and product-recovery section.
Three slide valves, located below the stripper, regenerator, and catalyst hopper, control catalyst-circulation rates. An online-gas analyzer with an infrared detector monitors the nitrogen, oxygen, carbon monoxide, and carbon dioxide contents in the flue gas from the regenerator. The product mass balance is typically more than 99%.
Except for its reactor system and catalyst hopper (no catalyst hopper in the riser pilot plant), the configuration of the conventional DCR riser unit is similar to that of the downer-pilot plant.
Table 1 shows typical reaction conditions of the two plants. The reactor outlet temperature of both pilot plants was 600° C.
Exchangers condense gaseous hydrocarbons from the stripper, and a stabilizer column separates the liquid hydrocarbons from the gases.
Liquid C5 products are cooled and batch distilled. A simulated-distillation gas chromatograph determined three liquid cuts: gasoline (C5-221° C.), LCO (light cycle oil, 221-343° C.), and HCO (heavy cycle oil, 343+° C.). Two gas chromatographs equipped with flame-ionization and thermal-conductivity detectors analyzed the gaseous products.
The experiments used hydrotreated Arabian Light vacuum gas oil (VGO) and a proprietary USY FCC catalyst specially designed for olefins maximization (supplied by Catalysts & Chemicals Ind. Co. Ltd., Kitaminato-Machi, Japan). Table 2 lists pertinent feed properties.
Table 3 presents the results of VGO cracking in both the downer and the riser reactor systems at 80 wt % conversion. Conversion is 100% less the weight percent of LCO and HCO.
Figs. 3a-3f plot product yields vs. conversion. For both systems, there were minor changes in the C/O ratio requirements and product selectivities.
In the downer system, conversion increased from 80 wt % to 83 wt % upon increasing the C/O ratio from 20 to 35 kg/kg. In the riser system, the conversion increased from 75 wt % to 81 wt % upon increasing the C/O ratio from 13 to 29 kg/kg (Fig. 3a).
At 80 wt % conversion, the downer required a C/O ratio of 20.0, and the riser required a C/O ratio of 29.4 kg/kg. The high reaction severity used in the experiments attributed to the reduced C/O ratio requirement.
In both systems, light olefins increased with increasing conversion (Fig. 3b). The riser showed higher yields of light olefins (mainly propylene) as a result of gasoline overcracking at 600° C. At 80 wt % conversion, the yields of light olefins were 23 wt % in the downer compared to 29 wt % in the riser. For propylene, the yield was about 8 wt % in the downer compared to 12 wt % in the riser.
Fig. 3c shows that as conversion increased, gasoline yields peaked then declined as a result of overcracking. The downer showed a 22% increase in gasoline yield compared to the riser system; the riser had a 39.9 wt % yield, and the downer had a 48.5 wt % yield. Higher selectivity of gasoline in the downer system is attributed to less back-mixing in the downer.8-9
Dry gas and 1,3-butadiene are typical products from thermal cracking of hydrocarbons (Figs. 3d-3e). These results indicate that thermal cracking of hydrocarbons is effectively suppressed in a downer reactor. This can be also explained by minimized back-mixing in the downer reactor.
Fig. 3f shows the coke yields. In both systems, coke increased with increased conversion. At 80 wt % conversion, coke yield in the downer was 2 wt %, compared to 3 wt % in the riser system.
For the other liquid products (LCO and HCO), both reactor systems yielded approximately the same amount: 11 wt % from the downer reactor and 9 wt % from the riser reactor.
Acknowledgment
The authors thank King Fahd University of Petroleum & Minerals, the Petroleum Energy Center, and MITI for their support of this work.
References
- Johnson, T., and Avidan, A., Hydrocarbon Tech. Intl., Spring 1994, p. 39.
- Fujiyama, Y., Process for fluid catalytic cracking of oils, US Patent No. 5,904,837, May 18, 1999.
- Letzsch, W., Petroleum Technology Quarterly, 1999, No. 2, p. 121.
- Jakkula, J., Niemi, J., and Lipianen, K., The Status of the Development of New Short Contact Time FCC Technology, presented at the 8th Saudi-Japanese Symposium on Catalysts in Petroleum Refining and Petrochemicals, KFUPM, Dhahran, November 1998.
- Chan, T., and Sundaram, K., SCC: Advanced FCCU Technology for Maximum Propylene Production, presented at the 2nd International Conference in Refinery Processing, AIChE Spring Meeting, Houston, March 1999.
- Eng, C., The Road to Propylene, MTO and PetroFCC, presented at UOP Petrochemicals Conference, Riyadh, January 2000.
- Ino, T., and Ikeda, S., Process for fluid catalytic cracking of heavy fraction oil, US Patent No. 5,951,850, Sept. 14, 1999.
- Ikeda, S., and Ino, T., Down-flow FCC pilot plant for light olefin production, presented at the 5th Intl. Symposium on Advances in FCC, 218th ACS National Meeting, New Orleans, August 1999.
- Takatsuka, T., Sato, S., Morimoto, Y., and Hashimoto, H., J., Japan Petroleum Institute, 1984, vol. 27, p. 533.
The authors-
Ali G. Maadhah is HS-FCC project manager and professor of chemical engineering at KFUPM. His research interests are FCC catalysis, petroleum refining, and petrochemicals. He has served as a manager of the petroleum and gas technology division at the university's Research Institute. Maadhah holds a PhD in chemical engineering from Oklahoma State University, Stillwater.
Mohammed Abul-Hamayel is an associate professor of chemical engineering at KFUPM. His research interests are catalysis, heat transfer, computer applications, and energy utilization. Previously, he was a director of the Information Technology Center at the university. Abul-Hamayel holds PhD in chemical engineering from Oklahoma State University.
Abdullah M. Aitani is a research scientist at the Center for Refining & Petrochemicals of the Research Institute at KFUPM. He is group leader for microactivity studies in the HS-FCC project. His research interests are applied catalysis, reaction engineering, and refining process applications. Aitani holds a PhD in chemistry from City University, London.
Takashi Ino is HS-FCC technical manager and senior researcher at the Petroleum Energy Center. He currently is a visiting senior researcher at the Center for Refining and Petrochemicals of the Research Institute at KFUPM. His research interests are applied catalysis and oil refining processes. Ino holds a PhD in chemistry from Waseda University, Tokyo.
Toshiaki Okuhara is working for the Petroleum Energy Center and is a visiting researcher at the Center for Refining & Petrochemicals of the Research Institute at KFUPM. His research interests are FCC catalysis and pilot plant operation. He has previously worked for Nippon Oil Co. Okuhara holds an MSc in chemistry from Waseda University.