Gas plant solves Hg problems with copper-sulfate absorbent
Based on a presentation to the 89th Annual GPA Convention, Mar. 21-24, 2010, Austin, Tex.
Operators at Saudi Aramco's Berri gas plant successfully resolved problems of high mercury content in the plant's feed gas by replacing activated carbon material with copper-sulfide mercury absorbent.
Moisture and performance issues were also resolved by adherence to appropriate unloading and loading procedures provided by the vendor in addition to gradually introducing gas to the bed. Outlet-mercury content analysis showed that copper sulfide has excellent mercury-absorbent capacity compared with activated carbon.
This article discusses the Berri gas plant's mercury-removal units' performance in 2007-08, bed replacement, commissioning, and operation experience.
Effects of mercury
Mercury is present in most produced natural gas streams and can have catastrophic effects on gas plants. At levels greater than 10 g/cu m, corrosion can damage aluminum heat exchangers due to mercury amalgamating with the aluminum, causing embrittlement, which reduces mechanical strength and results in unexpected failure and gas leakage.
The mercury also attacks low-temperature components in an LNG plant, poisons catalysts, and reduces the quality of refined products.
In addition, mercury and its compounds are extremely toxic to humans. They may enter the body by inhalation, ingestion, or penetration of the skin. Mercury can damage lungs, the central nervous system, and kidneys.
It may accumulate to such elevated levels that it adversely affects workers and the environment, particularly when pipelines and equipment are opened for maintenance or cleaning. Mercury is found in vapor and liquid—in condensate transport pipelines and process equipment—and also mixed with sludge.
Berri gas plant
The Berri gas plant is designed to process 1.42 bcfd of northern area associated production gases. BGP produces C3+ NGL, ethane, fuel and sales gas, and elemental sulfur. In addition, the plant currently produces energy for its power needs with provision to transport part of it to the Saudi Electrical Co.
Berri consists of the following units:
1. NGL recovery and gas processing.
2. Diglycolamine gas-sweetening.
3. Sulfur-recovery.
4. Ethane storage and handling.
5. Utility.
6. Sulfur pelletizing and exporting.
7. Sulfur-handling and storage.
8. Cogeneration plant.
9. Ethane-recovery plant.
Mercury-removal units are installed at the ethane-recovery plant (F-20), which includes two cryogenic turboexpander trains to recover a minimum 95% of the ethane in the residue gas from the NGL-recovery plant (470).
Plant-F20 operates three identical parallel reactors for removing mercury from natural gas to prevent stress corrosion cracking in the brazed aluminum exchangers. The combined low-pressure/high-pressure feed gas is fed through the mercury-removal units (D-630 A/B/C) to remove mercury to <10 g/cu m at the common outlet.
Each unit contains a charge of 72,300 lb (around 33 cu m) of copper sulfide, which removes any mercury in the feed gas (Fig. 1).
The mercury content of Plant F-20's feed streams was measured in 1998 at 30-40 g/cu m. Sulfur-impregnated carbon beds were therefore chosen because they are designed for inlet content of 100 g/cu m. The activated carbon is designed to remove mercury to less than 10 g/cu m.
After loading the activated carbon and commissioning MRU beds, gas plant operators worked to verify the performance of the beds and their capability to maintain <10 g/cu m mercury at the bed outlet.
Several mercury measurements were taken during 2005. All data showed that mercury content in the inlet exceeded 100 g/cu m. Consequently, mercury content in the outlet of the MRUs exceeded 10 g/cu m.
These results demonstrated that Plant F20's brazed aluminum heat exchangers were exposed to a mercury content in excess of the recommended 10 g/cu m level in the feed gas. Moreover, the feed gas to Plant-F20 was considerably higher than the design figure of the activated carbon beds (4 to 12 times the design value).
This high mercury content would increase the possibility of corrosion in the aluminum heat exchanger, resulting in a disaster. In addition, product consumers (petrochemical plants) will suffer from catalyst deactivation due to mercury in ethane and sales gas.
Operators evaluated several options to overcome the high mercury content in the feed gas:
• Option No. 1: In-kind replacement. This option was eliminated because the inlet feed's mercury concentration exceeded the design value of 100 g/cu m as feed inlet.
• Option No. 2: Silver-doped molecular sieve. The option calls for an extra layer of silver molecular sieve in the dehydrators to absorb the mercury. It was rejected because all mercury absorbed in the bed would be released in the sales gas grid during regeneration.
• Option No. 3: Sulfur-impregnated carbon. The Nucon material is a sulfur-impregnated carbon material with higher mercury-removal capacity than Calgon-impregnated carbon. This option was rejected because the material had not been tested as a replacement for the Calgon material.
• Option No. 4: Copper-sulfide material. Copper-sulfide material has a greater efficiency for mercury removal at higher inlet mercury concentrations. The material is a mixture of copper sulfide/basic copper carbonate, zinc sulfide/basic zinc carbonate, and aluminum oxide.
This material is designed to achieve lower than 10 g/cu m for an inlet feed gas of mercury content up to 100,000 g/cu m. The vendor claimed it had been tested in 50 different plants worldwide. The expected lifetime of this material would be about 25 years, with a minimum guaranteed life span of 5 years for an inlet mercury content of 100,000 g/cu m.
Based on the advantages of the copper sulfide, Berri plant operators decided to use it to replace the activated carbon in the MRUs.
First MRU bed replacement
In March 2006, the MRU vessels were loaded with copper sulfide and commissioned shortly thereafter. Fresh material is expected to last for at least 15 years based on 5,500 g/cu m mercury feed. Shortly after the beds were commissioned, catalyst performance dropped significantly.
Following completion of Bed C loading, the bed was leak tested by N2 and feed gas. Then, it was returned to service. The copper-sulfide material contains about 4% moisture.
Since the bed was not dried out before commissioning, this high amount of moisture resulted in hydrate formation in the chill down sections and the turboexpanders tripped several times. Moreover, methanol was frequently injected in several locations in the chilling sections to eliminate the high pressure drop across the heat exchangers.
The beds were changed out with fresh copper-sulfide material following a short purge with nitrogen. Since the restart, there have been major issues with hydrate formation on the cold parts of the plant.
Mercury measurements taken show that with an inlet mercury content of around 600 g/cu m, the initial levels leaving the beds were 20-80 g/cu m, with a combined outlet of around 25 g/cu m. As a result there were also concerns about the mercury-removal performance of the beds.
An investigation conducted by the vendor revealed that the cause of the low performance of the material appears to be the use of membrane-generated nitrogen during loading. This nitrogen contains 3% oxygen, which oxidizes the copper sulfide to copper sulfate (CuSO4).
Copper sulfate does not remove mercury and is also a desiccant. These two features reduced the mercury-removal performance and extended the drying time. Instead, 99.9% nitrogen should be used for blanketing vessels during loading of sulfided material.
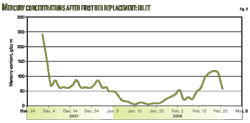
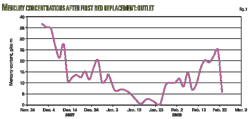
Berri plant operators worked to verify and evaluate the performance of the newly loaded copper-sulfide material. Analysis of mercury levels at the inlet and outlet and combined outlet positions were conducted for several months. Figs. 2 and 3 illustrate MRU inlet and outlet mercury analysis results from April 2006 to October 2007.
Bed premature failure
An investigation into the cause of the premature failure of the MRU beds revealed that the low performance of the material was mainly due to improper loading, which was caused by:
1. Using impure nitrogen (containing 3% oxygen) generated by Plant F-20's nitrogen membrane unit.
2. The high oxygen content oxidizes the copper sulfide to copper sulfate (CuSO4): CuS + 2O2 → CuSO4.
The material's exposure to high levels of oxygen resulted in its losing ability to remove mercury. The bed's efficiency was estimated to be less than 60%. As a substitute, 99.9% nitrogen should have been used for blanketing the vessels during loading the material to prevent this reaction.
Second bed replacement
The team recommended replacement of the entire old catalyst with fresh copper sulfide to maintain the safety and reliability of the downstream brazed aluminum heat exchanger.
Plant F-20 MRUs were shut down to replace the old catalyst with fresh copper-sulfide catalyst.
The key element in the loading procedure is the purity of the nitrogen used for purge. The oxygen content in purging nitrogen should be less than 0.1%; otherwise, oxygen would react with the copper sulfide to produce copper sulfate, as noted previously.
This reaction would degrade the performance of the catalyst because CuSO4 is not active for mercury removal. Therefore, during the purging stage, frequent checks and lab analyses were carried out to ensure that the vessel was under continuous (99.9%) N2 during bed loading. The nitrogen was supplied by Plant 476 because Plant F-20's nitrogen unit was unable to provide nitrogen with the required specification. Sampling every 2 hr confirmed the N2 purity.
The loading mechanism was as follows: Hopper and hoses were installed at the top manway and eight fresh absorbent drums were loaded into the hopper. As soon as an absorbent drum was opened, it was loaded into the hopper to the nitrogen-blanketed vessel. This allowed a limited material exposure to air. The hose was controlled by a contract operator with appropriate equipment and breathing apparatus in the vessel.
The main objective of this stage was to ensure that the catalyst not fall more than 1 m and cause damage or uneven distribution to the catalyst particles in the vessel.
Bed drying
Following completion of bed loading, the vessel was lined in preparation for the drying step. The objective of this step is to dispose of the moisture in the loaded copper-sulfide catalyst. The vendor states in the copper-sulfide certificate of analysis that the material contains an average of 2.7 wt % H2O. This is equivalent to 2,000 lb of water produced during each MRU bed commissioning. This massive amount of moisture must be dried out before bed commissioning to avoid operational upset/hydrate formation as the plant operates at low cryogenic temperatures.
1. Continuous flaring. As mentioned earlier, the copper-sulfide material contains about 2.7 wt % of moisture (equivalent to 1 ton/bed). Initially, the beds were dried out by introducing continuous flow through them and discharging it through the flare via a 2-in. line, which limited the flow to an average of 50 MMcfd. Drying gas was at 150° F. and 800 psi.
This methodology showed no significant improvement in the bed outlet's dew points. The dew point readings remained constant at the maximum range of the moisture analyzer (68° F.). The drying time of D-630B lasted more than 48 hr without progress.
Therefore, another methodology was proposed to accelerate drying.
2. Pressure swing. The purpose of this methodology is to accelerate bed drying to enable complete MRU bed replacement with the given timeframe. This method was implemented after 2 days of continuous drying the flare.
The bed was initially pressurized to 800 psi by low-pressure gas feed at 150° F., while the flaring flow was maintained at 50 MMcfd. After 2 hr of continuous flaring, the feed line was closed to depressurize the vessel to 0 psi. The depressurizing rate must be controlled to be lower than 2.5 psi/sec when vessel pressure is higher than 150 psi and 0.25 psi/sec when vessel pressure is lower than 150 psi.
This can be controlled manually with the available pressure gauge and valve opening at the discharge line. The depressurizing rate must not exceed the above limits to avoid shocking and fluidization of the bed material. This pressurizing-depressurizing step was repeated three times.
Pressure-swing cycle was repeated until the outlet dew point reaches zero.
Once the outlet dew point reached zero, the feed was gradually introduced to the system in increments of 50 MMcfd every 3 hr.
Fig. 4 shows the high differential pressure experienced in the heat exchangers and turboexpander, as a result of the moisture content escaping from the bed after being placed online.
With the pressure-swing method, operators were able to reduce the drying step to 5 days from 8 days. It can be noticed that drying efficiency is proportionally related to the drying gas flow rate and temperature. Therefore, the gas flow rate must be increased gradually (in small increments) once introduced to the plant to avoid high rate of moisture accumulation in the plant's cold boxes and turboexpanders. If high differential pressure appears on any of the plant's equipment, the feed gas flow rate must be adjusted until it is resolved.
Note
The material might generate some moisture if it has not been totally sulfided during manufacturing. One mole of hydrogen sulfide (H2S) in the feed gas will produce 1 mole of moisture across the MRU according the reaction stoichiometric indicated in the following: CuO + H2S → CuS + H2O.
The vendor confirmed that the material was not fully sulfided. Therefore, this reaction had contributed as well to the hydrate formations experienced.
Following the new beds' installation and commissioning, the performance of the loaded copper-sulfide catalyst was examined. The inlet and outlet mercury content of each bed was measured and analyzed. The results were satisfactory as the new material achieve the guaranteed value of 10 g/cu m. In fact, most of the outlet content readings were lower than 1 g/cu m.
MRU bed performance must be tested and verified while the inlet mercury content exceeds 100 g/cu m. This scenario would take place during certain modes of operation in the upstream facilities. It is subject to the production of oil wells and reservoir characterization for that well (i.e., pressure, flow, temperature).
Therefore, mercury content measurement will be continuous, once a week in the inlet and outlet of MRUs until completion of the installation of the online mercury analyzer at Plant F-20. Fig. 5 shows the results of mercury content measurements collected between August and October, 2008, after the installation of the new mercury beds.
The collected data showed that the beds successfully reduced outlet mercury concentrations leaving the MRUs to <10 g/cu m. The results showed that the mercury content in the common outlet of each bed was an average of 1.3 g/cu m.
The author
More Oil & Gas Journal Current Issue Articles
More Oil & Gas Journal Archives Issue Articles
View Oil and Gas Articles on PennEnergy.com